Is Liposomal NMN Worth It? What the Label Doesn't Tell You About Sunflower Lecithin
Most products labeled liposomal NMN have never been tested or verified to contain actual liposomes. Here is what the label is not telling you.

What to Know Before You Read
→ NMN is water-soluble. Water destroys liposomal structure. There is no format where both the molecule and the delivery system survive intact.
→ Most products labeled liposomal NMN have never been tested or verified to contain actual liposomes.
→ Real liposome manufacturing requires pharmaceutical-grade equipment, a defined process, and multiple validation steps. None of these are required in supplement production.
→ NMN has its own dedicated intestinal transporter, Slc12a8. It does not need a lipid delivery system.
→ Purity and crystalline structure are the only variables that actually determine NMN bioavailability. Enzymatic manufacturing produces the purest, most stable form. That is what every clinical trial used.
The Liposomal NMN Con
If you are researching NMN to support cellular health, DNA repair, and healthy aging, you have already seen the word everywhere. Liposomal. It is in the supplement name. It is on the homepage. The ads promise tiny fat bubbles that carry NMN directly into your cells, bypassing digestion and outperforming every other formula on the market. Worth paying 30 to 60 percent more for?
The con works because it borrows from real science. Liposomal technology genuinely exists. It is used in cancer treatment, IV drug delivery, and pharmaceutical research. The word carries weight because it earned weight, built over decades in settings with completely different equipment, standards, and costs.
What the supplement industry did was take that vocabulary and apply it to a dry powder capsule filled with sunflower lecithin. No pharmaceutical-grade equipment. No particle size validation. No encapsulation efficiency data. No proof that a single liposome exists in the product. Just the word. And the margin that comes with it.
Here is what you are actually buying: a supplement with sunflower lecithin added to the formula, a proprietary name applied to that lecithin, and marketing copy that connects both to pharmaceutical research that neither the ingredient nor the process resembles in any way.
Most of it is sunflower oil. And they are charging you like it is a pharmaceutical.
Where Liposomes Come From

The first liposomal drug the FDA approved was called Doxil. It came out in 1995. It was an oncology drug, toxic to the heart at high doses. Wrapping it in a lipid bilayer, a thin shell made of fat, allowed it to travel to tumor tissue without harming healthy cells along the way. Targeted delivery of a dangerous drug to a specific tissue. This is the gold standard of lipid-based drug delivery, and it required years of study with specialized equipment.
Building a real pharmaceutical liposome requires three ingredients, each measured to the milligram: a purified phospholipid, cholesterol, and a PEG chain. The phospholipid builds the wall. Cholesterol keeps it from falling apart. The PEG chain coats the outside, making the liposome invisible to the immune system. Without PEG, the body clears it before it reaches its target.
The lipids are dried into a thin film, frozen, thawed, frozen again, and forced through a filter ten times to produce particles of uniform size. Particle size must fall between 100 and 200 nanometers, roughly 500 times smaller than a human hair. Encapsulation efficiency must be above 70 percent to justify calling it a delivery system at all.
The supplement world saw this science. It borrowed the words. It skipped the work.
What Liposomal Actually Means on These Labels
Researchers have started testing these products. A nutraceuticals industry testing report found that most supplements claiming to be liposomal were not. Many had no liposomes at all. Some contained only micelles, simple clumps of fat molecules, far less sophisticated than a liposome. Most were nothing more than a mixture of sunflower lecithin and NMN with no liposome structures present of any kind.
A 2024 study testing multiple NMN products found that some liposomal versions delivered only 13% of their claimed dose. Others had no detectable NMN at all. The encapsulation may be trapping the molecule rather than delivering it.
There is no third-party program available to verify that liposomes exist in a product. A brand can print liposomal delivery on a label, mix some sunflower lecithin into a capsule, and face zero accountability.
The Cheap Ingredient With an Expensive Name

Look at the Other Ingredients panel of any supplement making a liposomal claim. You will see something like: phospholipids from non-GMO sunflower lecithin, including phosphatidylcholine.
Phosphatidylcholine is the dominant phospholipid naturally found in sunflower lecithin. They are the same thing. On most labels, the source is never disclosed. You see phosphatidylcholine, or phospholipid complex, with no mention of sunflower anywhere. The scientific name does the work of making a food-grade emulsifier sound like a pharmaceutical delivery system.
Sunflower lecithin is a byproduct of sunflower oil production, what is left over after sunflower seeds are pressed. It is a refined oilseed derivative. It is in your protein powder, your chocolate, your greens drink, your pre-workout, and now your NMN supplement. It replaced soy lecithin after soy became a liability. Allergy concerns, GMO debates, negative press. The shift had nothing to do with cellular health science.
The ingredient has a legitimate role as a manufacturing emulsifier. It helps oil and water mix. The same fatty substance that stops your protein shake from clumping is being positioned as a cellular transport mechanism. The chemistry did not change. The narrative did.
One more thing the longevity crowd deserves to know: sunflower lecithin carries residual linoleic acid, an omega-6 fat that breaks down easily when exposed to air, light, or heat. When it breaks down, it produces compounds linked to oxidative stress, the same kind of cellular damage most longevity buyers are trying to reduce. The people buying these products are often the same people who removed seed oils from their kitchen. The advanced delivery system in their capsule comes from the same oilseed source they have been trying to avoid.
Why NMN Doesn't Need Any of This
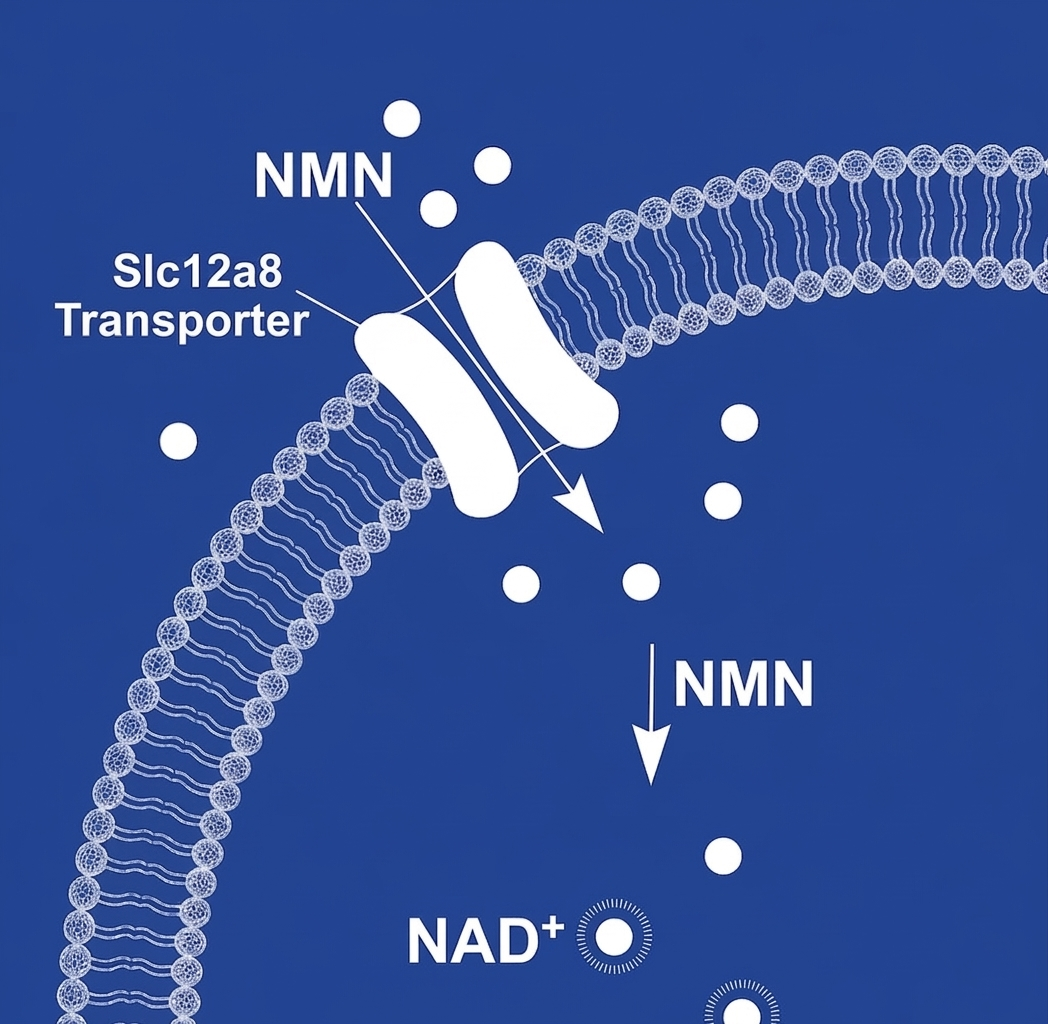
Liposomal delivery was developed for molecules with real absorption problems, compounds that break apart in the digestive system, struggle to cross the gut wall, or need to reach a specific tissue. NMN moves through the body on its own without those obstacles.
The small intestine has a dedicated transporter called Slc12a8 that pulls NMN directly into cells. It is a built-in uptake mechanism specific to this molecule. Liposomal encapsulation bypasses this transporter entirely, routing NMN through a different pathway that has never been shown to improve absorption in humans, and the available data suggests it may deliver less.
Even under ideal lab conditions, liposomal delivery of water-soluble compounds achieves only 20 to 40 percent efficiency. In dry powder capsules, that number is never tested or disclosed. A large portion of the NMN in these products may be floating free in the formula rather than enclosed in any delivery structure.
There is also a stability problem with no workaround. NMN degrades when it contacts water. Liposomes require water to hold their shape. In a dry capsule, liposome structures collapse. In a liquid format, the NMN begins breaking down. A stable product cannot satisfy both conditions at once.
What Pure Crystalline NMN Does Instead

Every human trial that demonstrated results, insulin sensitivity, aerobic capacity, muscle function, physical performance, used pharmaceutical-grade crystalline NMN. No liposomal delivery. No proprietary encapsulation. No sunflower lecithin.
A landmark study led by Yi L, Maier AB, Tao R and colleagues confirmed what earlier animal data suggested: oral crystalline NMN is well tolerated, raises NAD⁺ levels reliably, and converts to NAD⁺ in a single enzymatic step. That study used standard oral crystalline form.
The entire evidence base was built on the form the market is now telling you is inferior.
Purity is the variable that matters. Pharmaceutical-grade, enzymatically produced crystalline NMN has a dedicated transporter, a single conversion step to NAD⁺, and a growing body of human clinical trials behind it. It is stable at room temperature for over a year when kept dry. It needs to be what it is.
How to Read a Liposomal NMN Label Before You Buy
If the word liposomal is in the product name, go to the Other Ingredients panel first. If you see sunflower lecithin, sunflower seed extract, sunflower oil, soy lecithin, or phosphatidylcholine from sunflower, you are looking at a lecithin product. That is the complete story.
The same ingredient appears under many names:
Sunflower lecithin / Non-GMO sunflower lecithin / Sunflower seed lecithin / Purified sunflower seed lecithin / Phospholipids from sunflower lecithin / Phosphatidylcholine / Phosphatidylcholine from sunflower lecithin / Phospholipid complex / Soy lecithin
Every name on that list describes the same thing: a lecithin-derived phospholipid fraction from sunflower or soy. None constitute a validated liposomal delivery system.
Three questions worth asking any brand making this claim:
Can you provide dynamic light scattering data confirming particle size between 100 and 200 nanometers? If not, liposomes are unconfirmed.
Does your formulation include a PEG-lipid component? Without it, a defining feature of pharmaceutical liposomal technology is absent. No supplement product currently on the market includes one.
What is your encapsulation efficiency for NMN specifically? If they cannot provide this number, encapsulation is assumed, not confirmed.
The Bottom Line
The current evidence supports crystalline NMN. Multiple randomized, double-blind human trials have shown that oral crystalline NMN raises NAD⁺ levels, improves insulin sensitivity, supports metabolic health, and produces measurable effects on physical performance. The risk profile at standard doses is minimal.
Liposomal formats do not have equivalent data. They have a price premium, a marketing narrative, and a commodity ingredient.
Take the form the research was built on. Pharmaceutical-grade. Enzymatically produced. Third-party verified. Crystalline.
That is what the data supports.
Frequently Asked Questions
Is liposomal NMN better than regular NMN?
The evidence does not support that claim. Every human clinical trial demonstrating that NMN supplementation raises NAD+ levels, improves insulin sensitivity, supports physical performance, and enhances aerobic capacity used standard pharmaceutical-grade crystalline nicotinamide mononucleotide. No liposomal NMN supplement has ever been tested in a human clinical trial. The comparison being made in the marketing, liposomal versus regular NMN, has never been run as a study. One form has a clinical record built on double-blind, placebo-controlled research. The other has a price premium.
Do liposomal NMN supplements contain liposomes?
Independent testing says most do not. A nutraceuticals industry report found that the majority of supplements making liposomal claims could not be verified to contain liposomal structures of any kind. Some contained micelles. Others were simply a mixture of lecithin and NMN with nothing encapsulated. Liposomal encapsulation in a dry powder capsule is rarely confirmed and almost never published. There is no regulatory requirement and no third-party certification program that requires brands to prove liposomes exist before printing the word on a label.
What is the difference between liposomal NMN and crystalline NMN?
Crystalline NMN is a pure, stable, solid form of nicotinamide mononucleotide NMN produced through an enzymatic manufacturing process. It is the form used in every published human clinical trial on NMN supplementation. Liposomal NMN is NMN combined with a lecithin-based fatty substance, typically from sunflower seeds, under the assumption that a lipid delivery structure will form around the molecule. Independent testing has found that this structure rarely forms and, when it does, encapsulation efficiency is low. The practical difference for most buyers is that crystalline NMN has clinical trials behind it and liposomal NMN has a higher price.
What is phosphatidylcholine in NMN supplements?
Phosphatidylcholine is the dominant fatty substance found naturally in sunflower lecithin, derived from sunflower seeds. When it appears on an NMN supplement label, it is almost always from that source. It is not a separate pharmaceutical ingredient. It is not a validated liposomal delivery system. It is a component of a food-grade emulsifier found in chocolate, protein powders, and salad dressings. Listing it by its scientific name rather than its source creates the impression of something more advanced than it is. It is also worth noting that sunflower lecithin carries residual fatty acids, including linoleic acid, which oxidizes easily and contributes to the omega-6 imbalance many people taking NMN are already working to correct.
Does NMN need a special delivery system to be absorbed?
No. NMN is a naturally occurring molecule with a dedicated intestinal transporter called Slc12a8 that moves it directly into cells through the small intestine without any carrier system. This transporter was built specifically for nicotinamide mononucleotide. Liposomal delivery routes NMN around this pathway and through an alternative route used by fat-soluble compounds. NMN is not fat-soluble. It does not have the absorption problems that liposomal delivery was designed to solve. Research suggests bypassing the Slc12a8 pathway may reduce rather than improve how much NMN reaches cells.
Why does NMN degrade in liposomal products?
NMN is water-soluble and begins breaking down the moment it contacts water, even humidity inside a sealed capsule. Liposomes require water to maintain their structure. These two facts cannot coexist in a stable product. Remove the water and the liposomal structure collapses. Keep the water and the NMN degrades. Even in controlled animal models and laboratory settings, encapsulation efficiency for water-soluble compounds in liposomal preparations typically falls between 20 and 40 percent. Commercial products administered orally fare worse, and most never publish their encapsulation data. More research on liposomal delivery of water-soluble NAD+ precursors is needed, but the existing data does not favor it.
Is crystalline NMN stable at room temperature?
It depends on the form, and the difference is significant.
In water, NMN is not stable at room temperature. A 2023 degradation kinetics study found that NMN in aqueous solution loses roughly 10% of its potency in under four days at room temperature. That is not a storage problem. That is a formulation problem, and it is exactly why liquid and liposomal formats are compromised before they reach you.
Dry crystalline NMN behaves differently. Stability testing on pharmaceutical-grade crystalline powder stored under warm room conditions found an average purity loss of only 0.1% after six months. Under ideal conditions, properly packaged crystalline NMN has a shelf life of one to two years.
The variables that matter are moisture and heat, not temperature alone. Dry. Sealed. Cool. Dark. Those are the four conditions crystalline NMN needs to hold its potency. A liposomal format cannot satisfy the first requirement without compromising its own structure. Crystalline NMN satisfies all four without any delivery system at all.
One more thing worth knowing. Degraded NMN does not simply become less effective. It converts into nicotinamide, which at higher doses has been shown to inhibit sirtuins and PARP, a DNA repair enzyme, the exact pathways NMN is taken to support. What you are left with works against the reason you bought it.
Are there side effects from NMN supplements?
NMN is generally safe and well tolerated at standard doses across diverse populations. The GeroScience trial led by Yi L, Maier AB, Tao R and colleagues, which was randomized, double-blind, and placebo-controlled, found minimal risk and no serious adverse events in healthy middle-aged and older adults taking oral NMN over the study period. Some people report mild digestive system discomfort, including nausea or abdominal pain, particularly at higher doses. Long-term data in humans is still developing, and more studies are needed to fully characterize the safety profile of taking NMN over many years. If you have existing health concerns, including heart health conditions, cholesterol levels, or are managing LDL cholesterol, consult a healthcare professional or registered dietitian before starting.
Can NMN raise NAD+ levels?
Yes, according to multiple published human clinical trials. Nicotinamide adenine dinucleotide, or NAD+, declines significantly with age. The GeroScience trial confirmed that oral nicotinamide mononucleotide NMN supplementation reliably raises NAD+ levels in healthy middle-aged and older adults. Additional trials have shown positive effects on insulin sensitivity, aerobic capacity, and markers of metabolic health. Animal studies and animal models provided the early evidence base, and human trials have since confirmed the core biology. All of these trials used pharmaceutical-grade crystalline NMN administered orally. NAD+ decline is one of the most well-documented features of cellular aging, and NMN may be one of the few supplements with direct human evidence for raising it effectively.
What should I look for in a high-quality NMN supplement?Purity is the most important variable. Look for pharmaceutical-grade crystalline nicotinamide mononucleotide produced through an enzymatic process. Third-party testing should confirm identity, purity, and potency. A clean other ingredients panel matters. Foods found naturally to contain NMN, including broccoli, avocado, and cow's milk, contain only trace amounts, far below the doses used in clinical trials. Taking NMN supplements in crystalline form is the only way to reach the levels studied in human research. A product with a single active ingredient and no unnecessary additives is a product where the manufacturer is confident in the compound itself.
To deep dive on the best supplement to choose, check out this article.
What is enzymatic NMN and why does it matter?
Enzymatic NMN refers to nicotinamide mononucleotide produced through a biological enzymatic synthesis process rather than chemical synthesis. The enzymatic process produces a cleaner compound with fewer synthetic byproducts and a more consistent crystal structure. It is the production method associated with the highest purity NMN available and the method used by manufacturers supplying NMN for clinical research. When a brand specifies enzymatic manufacturing, it is describing how the compound was made, not just what it is.
Does NMN help with brain health or Alzheimer's disease?
Animal studies have shown promising results linking NAD+ levels to brain health, neuronal function, and markers associated with Alzheimer's disease and cognitive decline. Research suggests that raising NAD+ levels through NMN supplementation may support the cellular energy and DNA repair processes that decline in aging brain tissue. Human data in this area is still emerging and more studies are needed before conclusions can be drawn about NMN specifically for Alzheimer's disease or cognitive outcomes. The potential benefits are being actively studied, and the early signals from animal models are encouraging. What the current evidence supports is that NMN raises NAD+ levels in humans, and that NAD+ decline is associated with many features of brain aging. Whether NMN supplementation works to meaningfully slow that process in older adults is a question that ongoing clinical trials are designed to answer.
Is NMN found naturally in foods?
Yes. NMN is found in small amounts in foods including broccoli, edamame, avocado, cucumber, and cow's milk. However, the amounts found naturally in these foods are a fraction of the doses used in human clinical trials, typically 250 to 500 milligrams per day. Regular exercise also supports NAD+ levels through related pathways, but does not replace the direct precursor effect of taking NMN supplements at clinical doses. Food sources are a meaningful part of human health but are not a substitute for supplementation when the goal is to meaningfully raise NAD+ levels in the human body.
Key Takeaways
→ Most products labeled liposomal NMN have never been tested or verified to contain actual liposomes.
→ Real liposome manufacturing requires pharmaceutical-grade equipment, a defined process, and multiple validation steps. None of these are required in supplement production.
→ NMN is water-soluble. Water breaks down liposomal structure. There is no format where both the molecule and the delivery system survive intact.
→ NMN begins degrading the moment it contacts water. Even humidity in a bottle is enough. Liposomal NMN requires water to exist. Those two facts cannot coexist.
→ NMN has its own dedicated intestinal transporter, Slc12a8. It does not need a lipid delivery system.
→ Purity and crystalline structure are the only variables that actually determine NMN bioavailability. Enzymatic manufacturing produces the purest, most stable form. That is what every clinical trial used.
QUiET COYOTE Pure Crystalline NMN — One active ingredient. One step from NAD+. That is the point.